
Air volumetric and mass flowrate
Definition, Calculation and Conversions
Question or remark ? Please contact us at admin@powderprocess.net
| Section summary |
|---|
| 1. Definition of
airflow |
| 2. Calculation of air volumetric and mass flow |
| 3. Usage - Application |
1. Definition of airflow
The air flow is a fundamental data in pneumatic conveying systems. From the air flow, the air speed, solids loading ratio, pressure drop... can be calculated. It must however be noted that the air flow is usually given in normal conditions (Nm3/h) not necessary representative of the conditions in the conveying line as the air is compressible. The objective of this page is to show how to calculate the air volumetric flowrate at different conditions and calculate as well the mass flowrate.
2. Calculation of air volumetric and mass flow
How to calculate volumetric air flow ? How to calculate mass air flow ?
From the volumetric air flowrate given in conditions 1 (often the normal conditions, 101325 Pa and 20°c), the volumetric flow can be calculated in conditions 2. Conditions 2 should be either the beginning of the line or the end of the line.
The pressure in conveying line is not very high, few bar abs at maximum, thus the perfect gas law applies well.
Throughout the line, the number of moles of air / h is conserved, so the following equality can be written :

Equation 1 : calculation of the air volumetric flowrate at
different conditions
With :
- Q1 = air volumetric flow rate in (known) conditions (m3/h)- Q2 = air volumetric flow rate to be determined in conditions 2 (m3/h)
- P1 = pressure in conditions 1 (Pa)
- P2 = pressure in conditions 2 (Pa)
- T1 = temperature in conditions 1 (K)
- T2 = temperature in conditions 1 (K)
Top
5 Most Popular
1.
Pneumatic transport design guide
2. Ribbon
blenders
3. Powder mixing
4. Hoppers design guide
5. Measuring degree of
mixing
--------------
Top 5 New
1. Continuous Dry Mixing
2. Mixing speed
3. Mixer cycle time
optimization
4. Batch
/ continuous mixing comparison
5. Energy Savings
To calculate the air mass flowrate, it is necessary to calculate the air volumetric mass at the conditions studied. It can also be defined thanks to the perfect gas law.

With :
- ρair = volumetric mass of the air at the conditions considered (kg/m3)
- P = pressure at the conditions considered (Pa)
- T = temperature at the conditions considered (K)
- Mair = the molecular weight of air 0.029 kg/mol
The volumetric mass can thus be multiplied by the volumetric flowrate to get the air mass flowrate in the pneumatic conveying pipe :
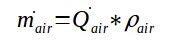
With :
- mair = air mass flowrate (kg/h)
- Qair = air volumetric flowrate at the conditions for which the volumetric mass has been calculated m3/h
ρair = volumetric mass of the air at the conditions considered (kg/m3)
3. Usage - Application
The following data can be calculated from the air volumetric flowrate and air mass flowrate
- Air conveying velocity at any point of the line
- Air solids ratio
- Pressure drop in pipes
Examples of calculation and conversions can be see on the page dedicated to the shortcut design method for calculation of dilute phase pneumatic conveying lines.